Preserving health
and quality of life

In today’s cancer therapy landscape, many cancer patients experience initial treatment success, leading to clinical remission. However, tumor recurrence remains an imminent threat and causes the vast majority of cancer-related deaths today. As a result, there is an increasing need for therapies that improve disease-free and overall survival following first-line treatment, particularly in tumor indications with a high recurrence rate.
Mendus is developing active immunotherapies, designed to stimulate the patient’s own immune system and establish long-term disease control without harming health or quality of life.
Addressing unmet needs in cancer treatment
Acute Myeloid leukemia (AML)
Disease relapse after initial treatment is the main barrier to long-term survival in AML, with measurable residual disease (MRD) being a key predictor of relapse and poor outcomes. Myeloid blood cancers like AML and CML are vulnerable to attack by the immune system. This principle has been shown extensively by hematopoietic stem cell transplantation (HSCT), a potentially curative but high-risk procedure associated with mortality and severe side effects including graft-versus-host disease.
Mendus is developing its lead product vididencel as an active immunotherapy to control residual disease and achieve durable clinical remissions in AML safely. This is particularly relevant for patients unable to undergo HSCT.
Chronic Myeloid Leukemia (CML)
CML, unlike AML, is a chronic disease that can be effectively controlled with targeted therapy based on tyrosine kinase inhibitors (TKIs). Thanks to the TKI success, overall survival expectations for CML patients today are close to the general population. However, the impact on quality of life and costs associated with continuous TKI treatment have led to a new frontier in the treatment of CML, which is to accomplish treatment-free remissions (TFR).
In CML, vididencel immunotherapy may improve TFR success, allowing more patients to live a healthy life without TKIs.
Ovarian Cancer
Ovarian cancer remains the deadliest gynecological malignancy, due to its high recurrence rate following first line surgery and chemotherapy. Vididencel has demonstrated tolerability and feasibility in high-risk ovarian cancer, providing a potential basis for novel combination treatments in this indication.

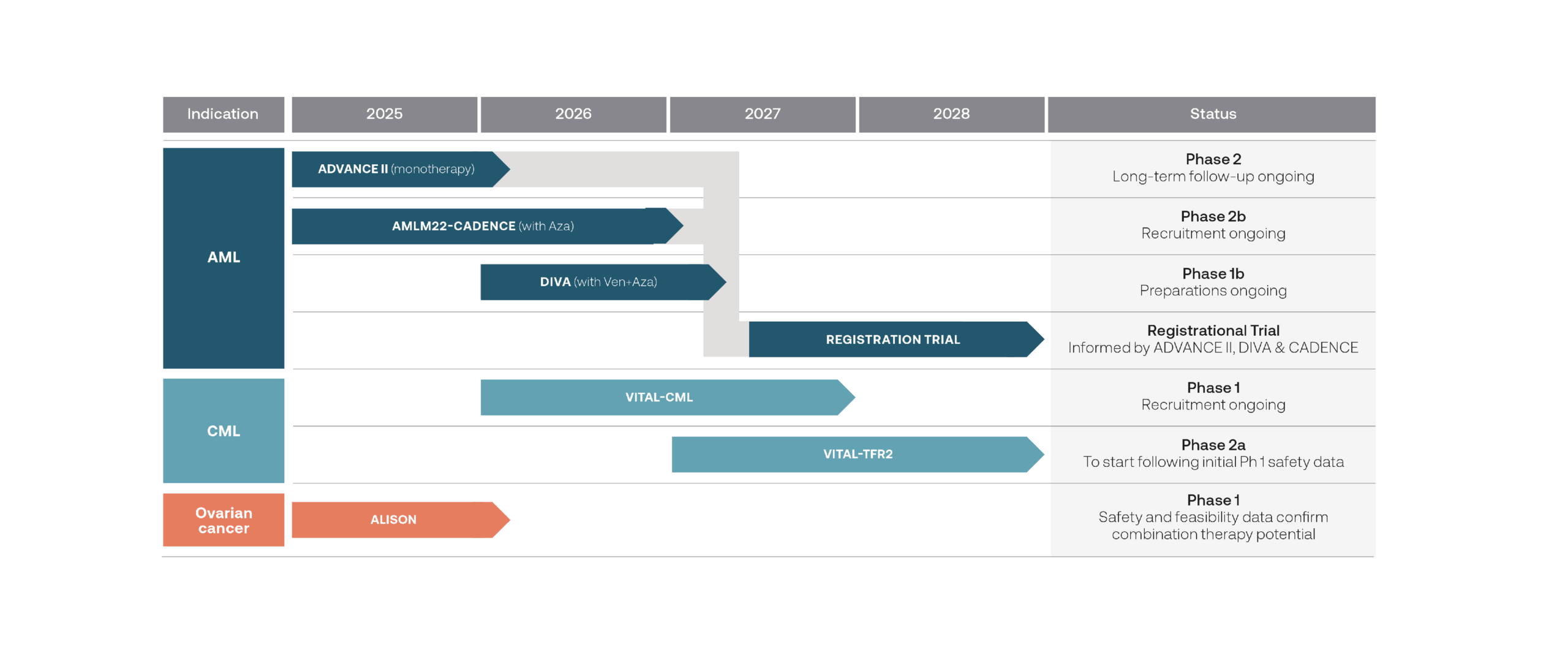
Pipeline
Technology
Dendritic cells play a central role in immune responses by training the immune system to recognize antigenic sequences produced by infections or tumor cells and by providing co-stimulatory signals that facilitate the activation and proliferation of T cells and other immune cells.

Vididencel
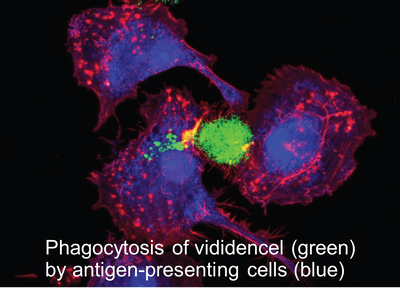
Vididencel is a cellular immunotherapy based on irradiated leukemic-derived dendritic cells. Following intradermal administration, vididencel induces a local immune response, activating antigen-presenting cells in the skin. These cells migrate to the lymph nodes, where they stimulate tumor-specific T-cell responses, supporting long-term immune control of residual disease.
DCOne platform
The DCOne platform supports the ex vivo expansion of immune cells with improved functionality for therapeutic purposes, including natural killer (NK) cells and tumor-infiltrating lymphocytes (TILs).
Publications
Patient Stories
Patient stories providing insight into medical needs, treatment experiences, and everyday challenges of living with cancer

Living with CML: Solveig’s Story
“This life I have on TKIs—it has a price. I don’t think it is a long-term solution, at least not for young people who cannot continue life the way they did before.”

Jacob’s AML journey
“Ann and I exercise every day together, taking walks in the forest or at the gym. We take this day by day and with our first grandchild set to join us in August we have a lot to look forward to.”

Giora Sharf’s 25-year history managing CML
“Don’t stop innovating—patients still need better, safer options.”

